AA74-1
AA74-1 (compound 23), completely inhibited APEH at 10 nM, while not interacting with other SHs at 100 nM.
General
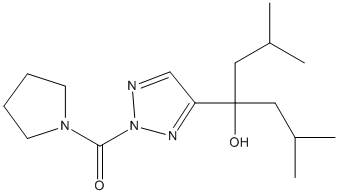
Type : Triazol,Pyrrolidine
Chemical_Nomenclature : [4-(4-hydroxy-2,6-dimethylheptan-4-yl)triazol-2-yl]-pyrrolidin-1-ylmethanone
Canonical SMILES : CC(C)CC(CC(C)C)(C1=NN(N=C1)C(=O)N2CCCC2)O
InChI : InChI=1S\/C16H28N4O2\/c1-12(2)9-16(22,10-13(3)4)14-11-17-20(18-14)15(21)19-7-5-6-8-19\/h11-13,22H,5-10H2,1-4H3
InChIKey : GXBVPGCIMCIASQ-UHFFFAOYSA-N
Other name(s) : (4-(4-Hydroxy-2,6-dimethylheptan-4-yl)-2H-1,2,3-triazol-2-yl)(pyrrolidin-1-yl)methanone,ZINC95099955,SR-02000001001
MW : 308.42
Formula : C16H28N4O2
CAS_number :
PubChem : 56593031
UniChem : GXBVPGCIMCIASQ-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
References (1)
| Title : Click-generated triazole ureas as ultrapotent in vivo-active serine hydrolase inhibitors - Adibekian_2011_Nat.Chem.Biol_7_469 |
| Author(s) : Adibekian A , Martin BR , Wang C , Hsu KL , Bachovchin DA , Niessen S , Hoover H , Cravatt BF |
| Ref : Nat Chemical Biology , 7 :469 , 2011 |
| Abstract : Adibekian_2011_Nat.Chem.Biol_7_469 |
| ESTHER : Adibekian_2011_Nat.Chem.Biol_7_469 |
| PubMedSearch : Adibekian_2011_Nat.Chem.Biol_7_469 |
| PubMedID: 21572424 |