B2Q
General
Type : Quinolizine
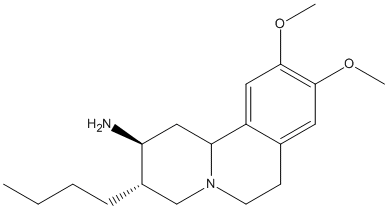
Chemical_Nomenclature : (2S,3S,11bS)-3-butyl-9,10-dimethoxy-2,3,4,6,7,11b-hexahydro-1H-benzo[a]quinolizin-2-amine
Canonical SMILES : CCCCC1CN2CCC3=CC(=C(C=C3C2CC1N)OC)OC
InChI : InChI=1S\/C19H30N2O2\/c1-4-5-6-14-12-21-8-7-13-9-18(22-2)19(23-3)10-15(13)17(21)11-16(14)20\/h9-10,14,16-17H,4-8,11-12,20H2,1-3H3\/t14-,16-,17-\/m0\/s1
InChIKey : IYTDSNSLAQHLJL-XIRDDKMYSA-N || IYTDSNSLAQHLJL-XIRDDKMYSA-P
Other name(s) : CHEMBL234412,BDBM50209938,(2S,3S,11bS)-3-butyl-9,10-dimethoxy-2,3,4,6,7,11b-hexahydro-1H-pyrido[2,1-a]isoquinolin-2-amine
MW : 318.46
Formula : C19H30N2O2
CAS_number :
UniChem : IYTDSNSLAQHLJL-XIRDDKMYSA-N || IYTDSNSLAQHLJL-XIRDDKMYSA-P
IUPHAR :
Wikipedia :
Target
Families : B2Q ligand of proteins in family: DPP4N_Peptidase_S9
Stucture : 3OC0 Structure of human DPP-IV with HTS hit (2S,3S,11bS)-3-Butyl-9,10-dimethoxy-1,3,4,6,7,11b-hexahydro-2H-pyrido[2,1-a]isoquinolin-2-ylamine (entry 3OC0 superseeds 3KWH 12-Jan-2010)
Protein : human-DPP4
References (1)
| Title : Discovery of carmegliptin: a potent and long-acting dipeptidyl peptidase IV inhibitor for the treatment of type 2 diabetes - Mattei_2010_Bioorg.Med.Chem.Lett_20_1109 |
| Author(s) : Mattei P , Boehringer M , Di Giorgio P , Fischer H , Hennig M , Huwyler J , Kocer B , Kuhn B , Loeffler BM , Macdonald A , Narquizian R , Rauber E , Sebokova E , Sprecher U |
| Ref : Bioorganic & Medicinal Chemistry Lett , 20 :1109 , 2010 |
| Abstract : Mattei_2010_Bioorg.Med.Chem.Lett_20_1109 |
| ESTHER : Mattei_2010_Bioorg.Med.Chem.Lett_20_1109 |
| PubMedSearch : Mattei_2010_Bioorg.Med.Chem.Lett_20_1109 |
| PubMedID: 20031405 |
| Gene_locus related to this paper: human-DPP4 |