Promazine
Chlorpromazine was the first antipsychotic drug to be discovered. It is one of the most sedating of the typical antipsychotics and also causes antimuscarinic effects.Tricyclic amine-containing compound. Promazine is a phenothiazine with actions similar to Chlorpromazine but with less antipsychotic activity. It is primarily used in short-term treatment of disturbed behavior and as an antiemetic.
General
Type : Phenothiazine,Drug,Sulfur Compound,Not A\/B H target
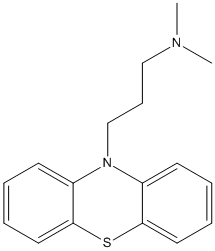
Chemical_Nomenclature : 10-[3-(dimethylamino)propyl]phenothiazine
Canonical SMILES : CN(C)CCCN1C2=CC=CC=C2SC3=CC=CC=C31
InChI : InChI=1S\/C17H20N2S\/c1-18(2)12-7-13-19-14-8-3-5-10-16(14)20-17-11-6-4-9-15(17)19\/h3-6,8-11H,7,12-13H2,1-2H3
InChIKey : ZGUGWUXLJSTTMA-UHFFFAOYSA-N
Other name(s) :
MW : 320.9
Formula : C17H20N2S
CAS_number : 53-60-1
PubChem : 4926
UniChem : ZGUGWUXLJSTTMA-UHFFFAOYSA-N
IUPHAR : 7281
Wikipedia : Promazine
Target
References (2)
| Title : Inhibition of butyrylcholinesterase by phenothiazine derivatives - Debord_2002_J.Enzyme.Inhib.Med.Chem_17_197 |
| Author(s) : Debord J , Merle L , Bollinger JC , Dantoine T |
| Ref : J Enzyme Inhib Med Chem , 17 :197 , 2002 |
| Abstract : Debord_2002_J.Enzyme.Inhib.Med.Chem_17_197 |
| ESTHER : Debord_2002_J.Enzyme.Inhib.Med.Chem_17_197 |
| PubMedSearch : Debord_2002_J.Enzyme.Inhib.Med.Chem_17_197 |
| PubMedID: 12443046 |
| Title : Three distinct domains in the cholinesterase molecule confer selectivity for acetyl- and butyrylcholinesterase inhibitors - Radic_1993_Biochemistry_32_12074 |
| Author(s) : Radic Z , Pickering NA , Vellom DC , Camp S , Taylor P |
| Ref : Biochemistry , 32 :12074 , 1993 |
| Abstract : Radic_1993_Biochemistry_32_12074 |
| ESTHER : Radic_1993_Biochemistry_32_12074 |
| PubMedSearch : Radic_1993_Biochemistry_32_12074 |
| PubMedID: 8218285 |