Amodiaquine
Amodiaquine is an orally active 4-aminoquinoline used for the therapy of malaria effective against some chloroquine-resistant strains of Plasmodium falciparum It is likely able to inhibit heme polymerase. Amodiaquine has been linked to severe cases of acute hepatitis and used only as treatment and not for prophylaxis against malaria. Amodiaquine shows mixed competitive-non-competitive inhibition type of AChE and shows slow and partial reactivation of sarin-, cyclosarin- and VX-inhibited cholinesterases. However it failed to reactivate tabun-inhibited human cholinesterases
General
Type : Quinoline,Not A\/B H target,Drug
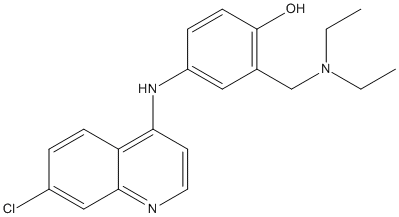
Chemical_Nomenclature : 4-[(7-chloroquinolin-4-yl)amino]-2-(diethylaminomethyl)phenol
Canonical SMILES : CCN(CC)CC1=C(C=CC(=C1)NC2=C3C=CC(=CC3=NC=C2)Cl)O
InChI : InChI=1S\/C20H22ClN3O\/c1-3-24(4-2)13-14-11-16(6-8-20(14)25)23-18-9-10-22-19-12-15(21)5-7-17(18)19\/h5-12,25H,3-4,13H2,1-2H3,(H,22,23)
InChIKey : OVCDSSHSILBFBN-UHFFFAOYSA-N
Other name(s) : ADQ,Amodiaquin,Camoquine,Flavoquine,Camoquin,Camochin
MW : 355.86
Formula : C20H22ClN3O
CAS_number : 86-42-0
PubChem : 2165
UniChem : OVCDSSHSILBFBN-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
References (2)
| Title : New therapeutic approaches and novel alternatives for organophosphate toxicity - Katz_2018_Toxicol.Lett_291_1 |
| Author(s) : Katz FS , Pecic S , Schneider L , Zhu Z , Hastings A , Luzac M , MacDonald J , Landry DW , Stojanovic MN |
| Ref : Toxicol Lett , 291 :1 , 2018 |
| Abstract : Katz_2018_Toxicol.Lett_291_1 |
| ESTHER : Katz_2018_Toxicol.Lett_291_1 |
| PubMedSearch : Katz_2018_Toxicol.Lett_291_1 |
| PubMedID: 29614332 |
| Title : Kinetic analysis of interactions of amodiaquine with human cholinesterases and organophosphorus compounds - Bierwisch_2016_Toxicol.Lett_246_49 |
| Author(s) : Bierwisch A , Wille T , Thiermann H , Worek F |
| Ref : Toxicol Lett , 246 :49 , 2016 |
| Abstract : Bierwisch_2016_Toxicol.Lett_246_49 |
| ESTHER : Bierwisch_2016_Toxicol.Lett_246_49 |
| PubMedSearch : Bierwisch_2016_Toxicol.Lett_246_49 |
| PubMedID: 26851641 |