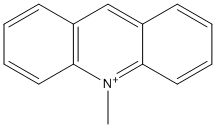
N-Methyl-acridinium
Tricyclic amine-containing compound
General
Type : Acridine
Chemical_Nomenclature : N-Methyl-acridinium
Canonical SMILES : C[N+]1=C2C=CC=CC2=CC3=CC=CC=C31
InChI : InChI=1S\/C14H12N\/c1-15-13-8-4-2-6-11(13)10-12-7-3-5-9-14(12)15\/h2-10H,1H3\/q+1
InChIKey : GXYLXFITCXXCQV-UHFFFAOYSA-N
Other name(s) : N-Methyl-acridine
MW : 194.26
Formula : C13H11N
CAS_number : 13367-81-2
PubChem : 22000
UniChem : GXYLXFITCXXCQV-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
References (2)
| Title : Binding of the neurotoxin fasciculin 2 to the acetylcholinesterase peripheral site drastically reduces the association and dissociation rate constants for N-methylacridinium binding to the active site - Rosenberry_1996_Biochemistry_35_685 |
| Author(s) : Rosenberry TL , Rabl CR , Neumann E |
| Ref : Biochemistry , 35 :685 , 1996 |
| Abstract : Rosenberry_1996_Biochemistry_35_685 |
| ESTHER : Rosenberry_1996_Biochemistry_35_685 |
| PubMedSearch : Rosenberry_1996_Biochemistry_35_685 |
| PubMedID: 8547248 |
| Title : Three distinct domains in the cholinesterase molecule confer selectivity for acetyl- and butyrylcholinesterase inhibitors - Radic_1993_Biochemistry_32_12074 |
| Author(s) : Radic Z , Pickering NA , Vellom DC , Camp S , Taylor P |
| Ref : Biochemistry , 32 :12074 , 1993 |
| Abstract : Radic_1993_Biochemistry_32_12074 |
| ESTHER : Radic_1993_Biochemistry_32_12074 |
| PubMedSearch : Radic_1993_Biochemistry_32_12074 |
| PubMedID: 8218285 |