Atglistatin
Specfic inhibitor of ATGL (Adipose Triglycerid lipase PNPLA2 Patatin-like phospholipase:not an alpha/beta hydrolase) Mayer et al. but inhibits also partially CES1 Iglesias et al 2016. ATGListatin inhibited mouse ATGL but not human ATG
General
Type : Urea derivative,Biphenyl
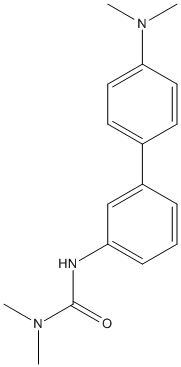
Chemical_Nomenclature : 3-[3-[4-(dimethylamino)phenyl]phenyl]-1,1-dimethylurea
Canonical SMILES : CN(C)C1=CC=C(C=C1)C2=CC(=CC=C2)NC(=O)N(C)C
InChI : InChI=1S\/C17H21N3O\/c1-19(2)16-10-8-13(9-11-16)14-6-5-7-15(12-14)18-17(21)20(3)4\/h5-12H,1-4H3,(H,18,21)
InChIKey : AWOPBSAJHCUSAS-UHFFFAOYSA-N
Other name(s) : 3-(4'-(Dimethylamino)-[1,1'-biphenyl]-3-yl)-1,1-dimethylurea,SCHEMBL15186954,AOB2795,MolPort-035-395-866
MW : 283.36
Formula : C17H21N3O
CAS_number : 1469924-27-3
PubChem : 71699712
UniChem : AWOPBSAJHCUSAS-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
References (3)
| Title : Unexpected role for IGF-1 in starvation: Maintenance of blood glucose - Fang_2022_Proc.Natl.Acad.Sci.U.S.A_119_e2208855119 |
| Author(s) : Fang F , Goldstein JL , Shi X , Liang G , Brown MS |
| Ref : Proc Natl Acad Sci U S A , 119 :e2208855119 , 2022 |
| Abstract : Fang_2022_Proc.Natl.Acad.Sci.U.S.A_119_e2208855119 |
| ESTHER : Fang_2022_Proc.Natl.Acad.Sci.U.S.A_119_e2208855119 |
| PubMedSearch : Fang_2022_Proc.Natl.Acad.Sci.U.S.A_119_e2208855119 |
| PubMedID: 35914126 |
| Title : Simplified assays of lipolysis enzymes for drug discovery and specificity assessment of known inhibitors - Iglesias_2016_J.Lipid.Res_57_131 |
| Author(s) : Iglesias J , Lamontagne J , Erb H , Gezzar S , Zhao S , Joly E , Truong VL , Skorey K , Crane S , Madiraju SR , Prentki M |
| Ref : J Lipid Res , 57 :131 , 2016 |
| Abstract : Iglesias_2016_J.Lipid.Res_57_131 |
| ESTHER : Iglesias_2016_J.Lipid.Res_57_131 |
| PubMedSearch : Iglesias_2016_J.Lipid.Res_57_131 |
| PubMedID: 26423520 |
| Title : Development of small-molecule inhibitors targeting adipose triglyceride lipase - Mayer_2013_Nat.Chem.Biol_9_785 |
| Author(s) : Mayer N , Schweiger M , Romauch M , Grabner GF , Eichmann TO , Fuchs E , Ivkovic J , Heier C , Mrak I , Lass A , Hofler G , Fledelius C , Zechner R , Zimmermann R , Breinbauer R |
| Ref : Nat Chemical Biology , 9 :785 , 2013 |
| Abstract : Mayer_2013_Nat.Chem.Biol_9_785 |
| ESTHER : Mayer_2013_Nat.Chem.Biol_9_785 |
| PubMedSearch : Mayer_2013_Nat.Chem.Biol_9_785 |
| PubMedID: 24096302 |