CHEMBL442384-S0E-3WK8
This fragment compound alos kills human pathogen Candida albicans by a specific inhibitor of non-essential mitotic kinesin Kip1p. || not really a good inhibitor IC50: 4.90e+4 (nM) using PHOME as substrate
General
Type : Benzothiazol,Not A\/B H target,Trifluoro,Sulfur Compound
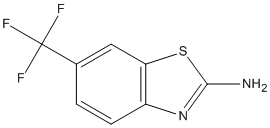
Chemical_Nomenclature : 6-(trifluoromethyl)-1,3-benzothiazol-2-amine
Canonical SMILES : C1=CC2=C(C=C1C(F)(F)F)SC(=N2)N
InChI : InChI=1S\/C8H5F3N2S\/c9-8(10,11)4-1-2-5-6(3-4)14-7(12)13-5\/h1-3H,(H2,12,13)
InChIKey : WEDYEBJLWMPPOK-UHFFFAOYSA-N
Other name(s) : 2-Amino-6-(trifluoromethyl)benzothiazole,6-(trifluoromethyl)benzo[d]thiazol-2-amine,2-amino-6-trifluoromethylbenzothiazole
MW : 218.20
Formula : C8H5F3N2S
CAS_number : 777-12-8
PubChem : 2735955
UniChem : WEDYEBJLWMPPOK-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
Families : CHEMBL442384-S0E-3WK8 ligand of proteins in family: Epoxide_hydrolase
Stucture : 3WK8 Crystal structure of soluble epoxide hydrolase in complex with fragment inhibitor 10
Protein : human-EPHX2
References (2)
| Title : Structural insights into binding of inhibitors to soluble epoxide hydrolase gained by fragment screening and X-ray crystallography - Amano_2014_Bioorg.Med.Chem_22_2427 |
| Author(s) : Amano Y , Yamaguchi T , Tanabe E |
| Ref : Bioorganic & Medicinal Chemistry , 22 :2427 , 2014 |
| Abstract : Amano_2014_Bioorg.Med.Chem_22_2427 |
| ESTHER : Amano_2014_Bioorg.Med.Chem_22_2427 |
| PubMedSearch : Amano_2014_Bioorg.Med.Chem_22_2427 |
| PubMedID: 24656800 |
| Gene_locus related to this paper: human-EPHX2 |
| Title : Effective killing of the human pathogen Candida albicans by a specific inhibitor of non-essential mitotic kinesin Kip1p - Chua_2007_Mol.Microbiol_65_347 |
| Author(s) : Chua PR , Roof DM , Lee Y , Sakowicz R , Clarke D , Pierce D , Stephens T , Hamilton M , Morgan B , Morgans D , Nakai T , Tomasi A , Maxon ME |
| Ref : Molecular Microbiology , 65 :347 , 2007 |
| Abstract : Chua_2007_Mol.Microbiol_65_347 |
| ESTHER : Chua_2007_Mol.Microbiol_65_347 |
| PubMedSearch : Chua_2007_Mol.Microbiol_65_347 |
| PubMedID: 17573815 |