Kurarinone
Kurarinone is a natural product found in Sophora flavescens, Sophora alopecuroides, Albizia julibrissin. Inhibits human-CES2 IC50 1.46+/-0.43, Ki 1.73 muM
General
Type : Natural,Flavonoid
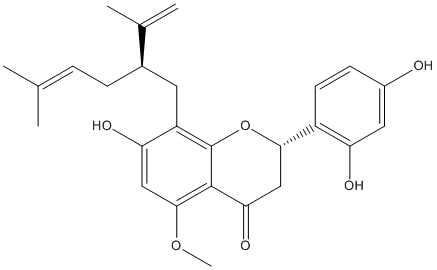
Chemical_Nomenclature : (2S)-2-(2,4-dihydroxyphenyl)-7-hydroxy-5-methoxy-8-(5-methyl-2-prop-1-en-2-ylhex-4-enyl)-2,3-dihydrochromen-4-one
Canonical SMILES : CC(=CCC(CC1=C2C(=C(C=C1O)OC)C(=O)CC(O2)C3=C(C=C(C=C3)O)O)C(=C)C)C
InChI : InChI=1S\/C26H30O6\/c1-14(2)6-7-16(15(3)4)10-19-21(29)12-24(31-5)25-22(30)13-23(32-26(19)25)18-9-8-17(27)11-20(18)28\/h6,8-9,11-12,16,23,27-29H,3,7,10,13H2,1-2,4-5H3\/t16?,23-\/m0\/s1
InChIKey : LTTQKYMNTNISSZ-KESSSICBSA-N
Other name(s) : (-)-kurarinone,CHEMBL243796,SCHEMBL10307523,BDBM50208612,(2S)-(-)-kurarinone,7,2',4'-Trihydroxy-8-lavandulyl-5-methoxyflavanone,CHEMBL492827,SCHEMBL904627,BDBM50486907
MW : 438.5
Formula : C26H30O6
CAS_number : 34981-26-5
PubChem : 10812923, 11982640, 5318882
UniChem : LTTQKYMNTNISSZ-KESSSICBSA-N
IUPHAR :
Wikipedia :
Target
Families : Kurarinone ligand of proteins in family: Carb_B_Chordata
Stucture : 7EBA Soluble epoxide hydrolase in complex with kurarinone
Protein : human-CES2
References (3)
| Title : Kurarinone alleviated Parkinson's disease via stabilization of epoxyeicosatrienoic acids in animal model - Sun_2022_Proc.Natl.Acad.Sci.U.S.A_119_ |
| Author(s) : Sun CP , Zhou JJ , Yu ZL , Huo XK , Zhang J , Morisseau C , Hammock BD , Ma XC |
| Ref : Proc Natl Acad Sci U S A , 119 : , 2022 |
| Abstract : Sun_2022_Proc.Natl.Acad.Sci.U.S.A_119_ |
| ESTHER : Sun_2022_Proc.Natl.Acad.Sci.U.S.A_119_ |
| PubMedSearch : Sun_2022_Proc.Natl.Acad.Sci.U.S.A_119_ |
| PubMedID: 35217618 |
| Gene_locus related to this paper: human-EPHX2 |
| Title : Five-Decade Update on Chemopreventive and Other Pharmacological Potential of Kurarinone: a Natural Flavanone - Kumar_2021_Front.Pharmacol_12_737137 |
| Author(s) : Kumar S , Prajapati KS , Shuaib M , Kushwaha PP , Tuli HS , Singh AK |
| Ref : Front Pharmacol , 12 :737137 , 2021 |
| Abstract : Kumar_2021_Front.Pharmacol_12_737137 |
| ESTHER : Kumar_2021_Front.Pharmacol_12_737137 |
| PubMedSearch : Kumar_2021_Front.Pharmacol_12_737137 |
| PubMedID: 34646138 |
| Title : Flavonoids as human carboxylesterase 2 inhibitors: Inhibition potentials and molecular docking simulations - Song_2019_Int.J.Biol.Macromol_131_201 |
| Author(s) : Song SS , Sun CP , Zhou JJ , Chu L |
| Ref : Int J Biol Macromol , 131 :201 , 2019 |
| Abstract : Song_2019_Int.J.Biol.Macromol_131_201 |
| ESTHER : Song_2019_Int.J.Biol.Macromol_131_201 |
| PubMedSearch : Song_2019_Int.J.Biol.Macromol_131_201 |
| PubMedID: 30872054 |