Naproxen-Ethyl-Ester
General
Type : Profen prodrug || Pro-Drug || Drug || Naphthalen || Propionate
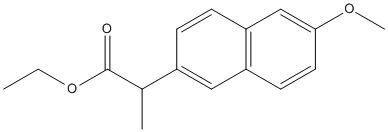
Chemical_Nomenclature : ethyl (2S)-2-(6-methoxynaphthalen-2-yl)propanoate
Canonical SMILES : CCOC(=O)C(C)C1=CC2=C(C=C1)C=C(C=C2)OC
InChI : InChI=1S\/C16H18O3\/c1-4-19-16(17)11(2)12-5-6-14-10-15(18-3)8-7-13(14)9-12\/h5-11H,4H2,1-3H3\/t11-\/m0\/s1
InChIKey : URNAYRDBUUZOIU-NSHDSACASA-N
Other name(s) : Naproxen Ethyl Ester, ethyl (2S)-2-(6-methoxynaphthalen-2-yl)propanoate, UNII-978RJD689Y, (S)-Naproxen Ethyl Ester, ZINC103450, SCHEMBL3280876
Target
Families : Canar_LipB, 6_AlphaBeta_hydrolase, Hormone-sensitive_lipase_like
References (4)
| Title : Enhanced Production of (S)-2-arylpropionic Acids by Protein Engineering and Whole-Cell Catalysis - Liu_2021_Front.Bioeng.Biotechnol_9_697677 |
| Author(s) : Liu X , Zhao M , Fan X , Fu Y |
| Ref : Front Bioeng Biotechnol , 9 :697677 , 2021 |
| Abstract : Liu_2021_Front.Bioeng.Biotechnol_9_697677 |
| ESTHER : Liu_2021_Front.Bioeng.Biotechnol_9_697677 |
| PubMedSearch : Liu_2021_Front.Bioeng.Biotechnol_9_697677 |
| PubMedID: 34307324 |
| Gene_locus related to this paper: 9zzzz-Est924 |
| Title : Crystal structures of two Bacillus carboxylesterases with different enantioselectivities - Rozeboom_2014_Biochim.Biophys.Acta_1844_567 |
| Author(s) : Rozeboom HJ , Godinho LF , Nardini M , Quax WJ , Dijkstra BW |
| Ref : Biochimica & Biophysica Acta , 1844 :567 , 2014 |
| Abstract : Rozeboom_2014_Biochim.Biophys.Acta_1844_567 |
| ESTHER : Rozeboom_2014_Biochim.Biophys.Acta_1844_567 |
| PubMedSearch : Rozeboom_2014_Biochim.Biophys.Acta_1844_567 |
| PubMedID: 24418394 |
| Gene_locus related to this paper: bacsu-cbxnp , bacsu-ybfk |
| Title : Lipase-catalyzed esterification of (S)-naproxen ethyl ester in supercritical carbon dioxide - Kwon_2009_J.Microbiol.Biotechnol_19_1596 |
| Author(s) : Kwon CH , Lee JH , Kim SW , Kang JW |
| Ref : J Microbiol Biotechnol , 19 :1596 , 2009 |
| Abstract : Kwon_2009_J.Microbiol.Biotechnol_19_1596 |
| ESTHER : Kwon_2009_J.Microbiol.Biotechnol_19_1596 |
| PubMedSearch : Kwon_2009_J.Microbiol.Biotechnol_19_1596 |
| PubMedID: 20075625 |
| Gene_locus related to this paper: canar-LipB |
| Title : Spherezymes: a novel structured self-immobilisation enzyme technology - Brady_2008_BMC.Biotechnol_8_8 |
| Author(s) : Brady D , Jordaan J , Simpson C , Chetty A , Arumugam C , Moolman FS |
| Ref : BMC Biotechnol , 8 :8 , 2008 |
| Abstract : Brady_2008_BMC.Biotechnol_8_8 |
| ESTHER : Brady_2008_BMC.Biotechnol_8_8 |
| PubMedSearch : Brady_2008_BMC.Biotechnol_8_8 |
| PubMedID: 18237402 |