Avenaol
General
Type : Strigolactone receptors ligand, Non-canonical strigolactone, Plant hormone
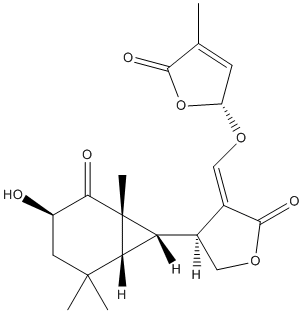
Chemical_Nomenclature : (2R)-2-[(Z)-[(4S)-4-[(1R,3R,6R,7S)-3-hydroxy-1,5,5-trimethyl-2-oxo-7-bicyclo[4.1.0]heptanyl]-2-oxooxolan-3-ylidene]methoxy]-4-methyl-2H-furan-5-one
Canonical SMILES : CC1=CC(OC1=O)OC=C2C(COC2=O)C3C4C3(C(=O)C(CC4(C)C)O)C
InChI : InChI=1S\/C20H24O7\/c1-9-5-13(27-17(9)23)25-8-11-10(7-26-18(11)24)14-15-19(2,3)6-12(21)16(22)20(14,15)4\/h5,8,10,12-15,21H,6-7H2,1-4H3\/b11-8-\/t10-,12-,13-,14-,15-,20-\/m1\/s1
InChIKey : MZPHYCYAMMTPLA-VUFGVBPYSA-N
Other name(s) : (+)-Avenaol
MW : 376.4
Formula : C20H24O7
CAS_number :
PubChem : 102236949
UniChem : MZPHYCYAMMTPLA-VUFGVBPYSA-N
Target
Families : Avenaol ligand of proteins in family
RsbQ-like
References (4)
| Title : Identification of 6-epi-heliolactone as a biosynthetic precursor of avenaol in Avena strigosa - Moriyama_2022_Biosci.Biotechnol.Biochem_86_998 |
| Author(s) : Moriyama D , Wakabayashi T , Shiotani N , Yamamoto S , Furusato Y , Yabe K , Mizutani M , Takikawa H , Sugimoto Y |
| Ref : Biosci Biotechnol Biochem , 86 :998 , 2022 |
| Abstract : |
| PubMedSearch : Moriyama_2022_Biosci.Biotechnol.Biochem_86_998 |
| PubMedID: 35561745 |
| Title : Molecular Transformation Based on an Innovative Catalytic System - Takemoto_2021_Chem.Pharm.Bull.(Tokyo)_69_819 |
| Author(s) : Takemoto Y |
| Ref : Chem Pharm Bull (Tokyo) , 69 :819 , 2021 |
| Abstract : |
| PubMedSearch : Takemoto_2021_Chem.Pharm.Bull.(Tokyo)_69_819 |
| PubMedID: 34470946 |
| Title : Lotuslactone, a non-canonical strigolactone from Lotus japonicus - Xie_2019_Phytochemistry_157_200 |
| Author(s) : Xie X , Mori N , Yoneyama K , Nomura T , Uchida K , Akiyama K |
| Ref : Phytochemistry , 157 :200 , 2019 |
| Abstract : |
| PubMedSearch : Xie_2019_Phytochemistry_157_200 |
| PubMedID: 30439621 |
| Title : [Construction of All-cis-substituted Cyclopropane and Total Synthesis of Avenaol] - Yasui_2019_Yakugaku.Zasshi_139_1259 |
| Author(s) : Yasui M |
| Ref : Yakugaku Zasshi , 139 :1259 , 2019 |
| Abstract : |
| PubMedSearch : Yasui_2019_Yakugaku.Zasshi_139_1259 |
| PubMedID: 31582609 |