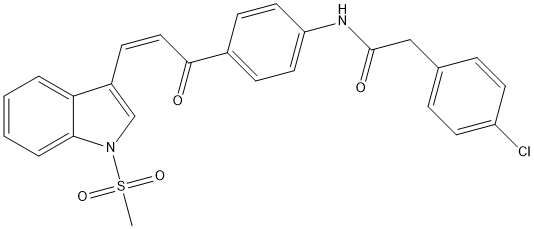
Chlorophenyl-methylsulfonyl-indol-acryloyl-phenyl-acetamide-cpd5b
AChE IC50 = 27.54nM BuChE IC50 = 36.85nM
General
Type : Multitarget,Indole,Carboxamide,Sulfur Compound,Sulfonamide
Chemical_Nomenclature : (E)-2(4-Chlorophenyl)-N-{4-[3(1-(methylsulfonyl)-1H-indol-3-yl)acryloyl]phenyl}acetamide
Canonical SMILES : C1=CC=C2C(=C1)C(=C[N]2[S](=O)(=O)C)C=CC(=O)C3=CC=C(C=C3)NC(=O)CC4=CC=C(C=C4)Cl
InChI : InChI=1S\/C26H21ClN2O4S\/c1-34(32,33)29-17-20(23-4-2-3-5-24(23)29)10-15-25(30)19-8-13-22(14-9-19)28-26(31)16-18-6-11-21(27)12-7-18\/h2-15,17H,16H2,1H3,(H,28,31)\/b15-10+
InChIKey : BBEIFPFQIPYISC-XNTDXEJSSA-N
Other name(s) : Compound 5b
MW : 492.97
Formula : C26H21ClN2O4S
CAS_number :
PubChem :
UniChem : BBEIFPFQIPYISC-XNTDXEJSSA-N
IUPHAR :
Wikipedia :
Target
Families : Chlorophenyl-methylsulfonyl-indol-acryloyl-phenyl-acetamide-cpd5b ligand of proteins in family: BCHE || ACHE
Stucture :
Protein : human-BCHE || human-ACHE
References (1)
| Title : Design and synthesis of new indole drug candidates to treat Alzheimer's disease and targeting neuro-inflammation using a multi-target-directed ligand (MTDL) strategy - Lamie_2022_J.Enzyme.Inhib.Med.Chem_37_2660 |
| Author(s) : Lamie PF , Abdel-Fattah MM , Philoppes JN |
| Ref : J Enzyme Inhib Med Chem , 37 :2660 , 2022 |
| Abstract : Lamie_2022_J.Enzyme.Inhib.Med.Chem_37_2660 |
| ESTHER : Lamie_2022_J.Enzyme.Inhib.Med.Chem_37_2660 |
| PubMedSearch : Lamie_2022_J.Enzyme.Inhib.Med.Chem_37_2660 |
| PubMedID: 36146947 |