Nu-1250
General
Type : Carbamate,Trimethylammonium
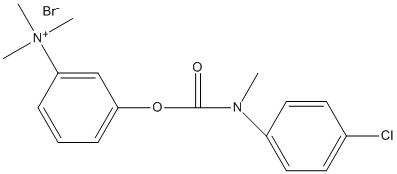
Chemical_Nomenclature : N-p-chlorophenyl-N-methylcarbamate of m-hydroxyphenyltrimethylammonium bromide
Canonical SMILES : C1=C(C=CC=C1OC(N(C)C2=CC=C(C=C2)Cl)=O)[N+](C)(C)C.[Br-]
InChI : InChI=1S\/C17H20ClN2O2.BrH\/c1-19(14-10-8-13(18)9-11-14)17(21)22-16-7-5-6-15(12-16)20(2,3)4\;\/h5-12H,1-4H3\;1H\/q+1\;\/p-1
InChIKey : PCQGALCVXJWHNH-UHFFFAOYSA-M
Other name(s) :
Target
Families : Nu-1250 ligand of proteins in family
ACHE
References (3)
| Title : [Anticholinesteratic and pharmacological effects of the bromide of N-P-Chlorophenyl-methylcarbamate of M-Hydroxyphenyltrimethylammonium (NU 1250)] - |
| Author(s) : Casier H , De Vleeschhouwer GR |
| Ref : Archives Internationales de Pharmacodynamie et de Therapie , 90 :412 , 1952 |
| PubMedID: 12986938 |
| Title : Tubocurarine antagonism and inhibition of cholinesterases - |
| Author(s) : Blaschko H , Bulbring E , Chou TC |
| Ref : British Journal of Pharmacology , 4 :29 , 1949 |
| PubMedID: 18113150 |