tacrine-isatin-Cpd7k
IC50 values of 0.42nM
General
Type : anti-Abeta-aggregation,Derivative of Tacrine,Multitarget,Alkyl linked bis-ligand,Indole
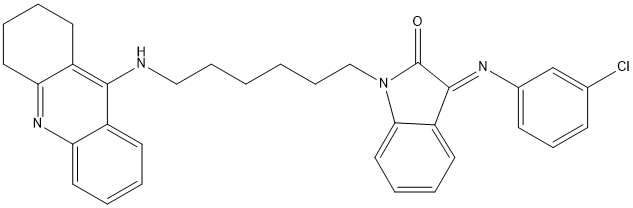
Chemical_Nomenclature : 3-((3-chlorophenyl)imino)-1-(6-((1,2,3,4-tetrahydroacridin-9-yl)amino)hexyl)indolin-2-one
Canonical SMILES : C3CCC2=NC1=[C][C]=[C][C]=C1C(=C2C3)[N]CCCCCCN5C4=[C][C]=[C][C]=C4C(C5=O)=NC6=[C][C]=[C]C(=[C]6)Cl
InChI : InChI=1S\/C33H33ClN4O\/c34-23-12-11-13-24(22-23)36-32-27-16-5-8-19-30(27)38(33(32)39)21-10-2-1-9-20-35-31-25-14-3-6-17-28(25)37-29-18-7-4-15-26(29)31\/h3,5-6,8,11-14,16-17,19,22H,1-2,4,7,9-10,15,18,20-21H2,(H,35,37)
InChIKey : HJOYCIDPPBFLLR-UHFFFAOYSA-N
Other name(s) :
MW : 537.10
Formula : C33H33ClN4O
CAS_number :
PubChem :
UniChem : HJOYCIDPPBFLLR-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
References (1)
| Title : Design, synthesis and biological activity of novel tacrine-isatin Schiff base hybrid derivatives - Riazimontazer_2019_Bioorg.Chem_89_103006 |
| Author(s) : Riazimontazer E , Sadeghpour H , Nadri H , Sakhteman A , Tuylu Kucukkilinc T , Miri R , Edraki N |
| Ref : Bioorg Chem , 89 :103006 , 2019 |
| Abstract : Riazimontazer_2019_Bioorg.Chem_89_103006 |
| ESTHER : Riazimontazer_2019_Bioorg.Chem_89_103006 |
| PubMedSearch : Riazimontazer_2019_Bioorg.Chem_89_103006 |
| PubMedID: 31158577 |