Glycolate
glycolate product of hydrolysis of Fluoroacetate
General
Type : Acetate
Chemical_Nomenclature : 2-hydroxyacetic acid
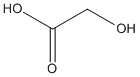
Canonical SMILES : C(C(=O)O)O
InChI : InChI=1S\/C2H4O3\/c3-1-2(4)5\/h3H,1H2,(H,4,5)
InChIKey : AEMRFAOFKBGASW-UHFFFAOYSA-N || AEMRFAOFKBGASW-UHFFFAOYSA-M
Other name(s) : GOA,Glycolic acid,2-Hydroxyacetic acid,Hydroxyacetic acid,79-14-1,Glycollic acid,Hydroxyethanoic acid,Hydroxyacetate,2-hydroxyacetate,2-oxidanylethanoate,AC1NUT2I,CHEBI:29805
MW : 76.05
Formula : C2H4O3
CAS_number :
UniChem : AEMRFAOFKBGASW-UHFFFAOYSA-N || AEMRFAOFKBGASW-UHFFFAOYSA-M
IUPHAR :
Wikipedia :
Target
Families : Glycolate ligand of proteins in family: Haloacetate_dehalogenase
Stucture :
Protein : rhopa-q6nam1
References (4)
| Title : Time-resolved crystallography reveals allosteric communication aligned with molecular breathing - Mehrabi_2019_Science_365_1167 |
| Author(s) : Mehrabi P , Schulz EC , Dsouza R , Muller-Werkmeister HM , Tellkamp F , Miller RJD , Pai EF |
| Ref : Science , 365 :1167 , 2019 |
| Abstract : Mehrabi_2019_Science_365_1167 |
| ESTHER : Mehrabi_2019_Science_365_1167 |
| PubMedSearch : Mehrabi_2019_Science_365_1167 |
| PubMedID: 31515393 |
| Gene_locus related to this paper: rhopa-q6nam1 |
| Title : Substrate-Based Allosteric Regulation of a Homodimeric Enzyme - Mehrabi_2019_J.Am.Chem.Soc_141_11540 |
| Author(s) : Mehrabi P , Di Pietrantonio C , Kim TH , Sljoka A , Taverner K , Ing C , Kruglyak N , Pomes R , Pai EF , Prosser RS |
| Ref : Journal of the American Chemical Society , 141 :11540 , 2019 |
| Abstract : Mehrabi_2019_J.Am.Chem.Soc_141_11540 |
| ESTHER : Mehrabi_2019_J.Am.Chem.Soc_141_11540 |
| PubMedSearch : Mehrabi_2019_J.Am.Chem.Soc_141_11540 |
| PubMedID: 31188575 |
| Gene_locus related to this paper: rhopa-q6nam1 |
| Title : The role of dimer asymmetry and protomer dynamics in enzyme catalysis - Kim_2017_Science_355_ |
| Author(s) : Kim TH , Mehrabi P , Ren Z , Sljoka A , Ing C , Bezginov A , Ye L , Pomes R , Prosser RS , Pai EF |
| Ref : Science , 355 : , 2017 |
| Abstract : Kim_2017_Science_355_ |
| ESTHER : Kim_2017_Science_355_ |
| PubMedSearch : Kim_2017_Science_355_ |
| PubMedID: 28104837 |
| Gene_locus related to this paper: rhopa-q6nam1 |
| Title : Mapping the reaction coordinates of enzymatic defluorination - Chan_2011_J.Am.Chem.Soc_133_7461 |
| Author(s) : Chan PW , Yakunin AF , Edwards EA , Pai EF |
| Ref : Journal of the American Chemical Society , 133 :7461 , 2011 |
| Abstract : Chan_2011_J.Am.Chem.Soc_133_7461 |
| ESTHER : Chan_2011_J.Am.Chem.Soc_133_7461 |
| PubMedSearch : Chan_2011_J.Am.Chem.Soc_133_7461 |
| PubMedID: 21510690 |
| Gene_locus related to this paper: rhopa-q6nam1 |