pro-2PAM
General
Type : Oxime, Pyridine-aldoxime, Organochlorine
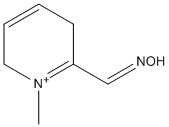
Chemical_Nomenclature : N-methyl-1,6-dihydropyridine-2-carbaldoxime hydrochloride
Canonical SMILES : CN1CC=CC=C1C=NO.Cl
InChI : InChI=1S\/C7H10N2O.ClH\/c1-9-5-3-2-4-7(9)6-8-10\;\/h2-4,6,10H,5H2,1H3\;1H\/b8-6+\;
InChIKey : REIIOHUYMPSAPS-WVLIHFOGSA-N
Other name(s) : Pro-pam || 1-methyl-1,6-dihydropyridine-2-carbaldoxime
MW : 174.628
Formula : C7H11ClN2O
CAS_number :
PubChem : 9587901
UniChem : REIIOHUYMPSAPS-WVLIHFOGSA-N
Target
Structure : No structure
Families : No family
References (3)
| Title : Amidine-oximes: reactivators for organophosphate exposure - Kalisiak_2011_J.Med.Chem_54_3319 |
| Author(s) : Kalisiak J , Ralph EC , Zhang J , Cashman JR |
| Ref : Journal of Medicinal Chemistry , 54 :3319 , 2011 |
| Abstract : |
| PubMedSearch : Kalisiak_2011_J.Med.Chem_54_3319 |
| PubMedID: 21438612 |
| Title : Reactivating and protective effects of Pro-2PAM in mice poisoned with paraoxon - |
| Author(s) : Boskovic B , Tadic V , Kusic R |
| Ref : Toxicol Appl Pharmacol , 55 :32 , 1980 |
| PubMedID: 7423504 |
| Title : Improved delivery through biological membranes. 3. Delivery of N-methylpyridinium-2-carbaldoxime chloride through the blood-brain barrier in its dihydropyridine pro-drug form - Shek_1976_J.Med.Chem_19_113 |
| Author(s) : Shek E , Higuchi T , Bodor N |
| Ref : Journal of Medicinal Chemistry , 19 :113 , 1976 |
| Abstract : |
| PubMedSearch : Shek_1976_J.Med.Chem_19_113 |
| PubMedID: 1246032 |