IJJ
Pks13 IC50 0.3 microM, H37Rv MIC 11microM
General
Type : Carboxamide,Oxadiazol,Azetidin
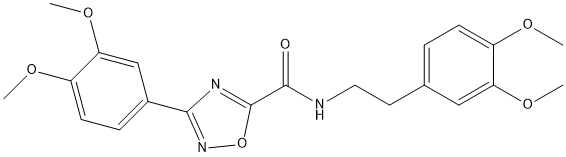
Chemical_Nomenclature : 3-(3,4-dimethoxyphenyl)-N-[2-(3,4-dimethoxyphenyl)ethyl]-1,2,4-oxadiazole-5-carboxamide
Canonical SMILES : COC1=C(C=C(C=C1)CCNC(=O)C2=NC(=NO2)C3=CC(=C(C=C3)OC)OC)OC
InChI : InChI=1S\/C21H23N3O6\/c1-26-15-7-5-13(11-17(15)28-3)9-10-22-20(25)21-23-19(24-30-21)14-6-8-16(27-2)18(12-14)29-4\/h5-8,11-12H,9-10H2,1-4H3,(H,22,25)
InChIKey : GVKZOYXTOVFRQE-UHFFFAOYSA-N
Other name(s) : Compound 50,CHEMBL2097844,TCMDC-142409,GSK796865A,AKOS001739861
MW : 413.4
Formula : C21H23N3O6
CAS_number :
PubChem : 1508946
UniChem : GVKZOYXTOVFRQE-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
Families : IJJ ligand of proteins in family: Thioesterase
Stucture : 8Q0T Mycobacterium tuberculosis Polyketide synthetase 13 thioesterase domain in complex with IJJ Compound 50 with antitubercular activity
Protein : myctu-PKS13
References (1)
| Title : Identification and Optimization of Novel Inhibitors of the Polyketide Synthase 13 Thioesterase Domain with Antitubercular Activity - Green_2023_J.Med.Chem__ |
| Author(s) : Green SR , Wilson C , Eadsforth TC , Punekar AS , Tamaki FK , Wood G , Caldwell N , Forte B , Norcross NR , Kiczun M , Post JM , Lopez-Roman EM , Engelhart CA , Lukac I , Zuccotto F , Epemolu O , Boshoff HIM , Schnappinger D , Walpole C , Gilbert IH , Read KD , Wyatt PG , Baragana B |
| Ref : Journal of Medicinal Chemistry , : , 2023 |
| Abstract : Green_2023_J.Med.Chem__ |
| ESTHER : Green_2023_J.Med.Chem__ |
| PubMedSearch : Green_2023_J.Med.Chem__ |
| PubMedID: 37948640 |
| Gene_locus related to this paper: myctu-PKS13 |