SCHEMBL1719815
General
Type : Cyanide, Sulfur Compound, Thiadiazol
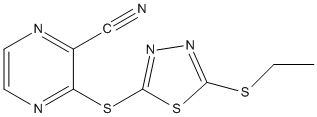
Chemical_Nomenclature : 3-[(5-ethylsulfanyl-1,3,4-thiadiazol-2-yl)sulfanyl]pyrazine-2-carbonitrile
Canonical SMILES : CCSC1=NN=C(S1)SC2=NC=CN=C2C#N
InChI : InChI=1S\/C9H7N5S3\/c1-2-15-8-13-14-9(17-8)16-7-6(5-10)11-3-4-12-7\/h3-4H,2H2,1H3
InChIKey : UBSFSJMIJHSGDZ-UHFFFAOYSA-N
Other name(s) : ZINC116926570 || LCAT activator compound A, >=98\% (HPLC)' || 3-(5-(ethylthio)-1,3,4-thiadiazol-2-ylthio)pyrazine-2-carbonitrile || 3-[(5-ethylsulfanyl-1,3,4-thiadiazol-2-yl)sulfanyl]pyrazine-2-carbonitrile
MW : 281.4
Formula : C9H7N5S3
CAS_number :
PubChem : 23642189
UniChem : UBSFSJMIJHSGDZ-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
Families : PC-sterol_acyltransferase
Protein : human-LCAT
References (2)
| Title : Lecithin:Cholesterol Acyltransferase Activation by Sulfhydryl-Reactive Small Molecules: Role of Cysteine-31 - Freeman_2017_J.Pharmacol.Exp.Ther_362_306 |
| Author(s) : Freeman LA , Demosky SJ, Jr. , Konaklieva M , Kuskovsky R , Aponte A , Ossoli AF , Gordon SM , Koby RF , Manthei KA , Shen M , Vaisman BL , Shamburek RD , Jadhav A , Calabresi L , Gucek M , Tesmer JJG , Levine RL , Remaley AT |
| Ref : Journal of Pharmacology & Experimental Therapeutics , 362 :306 , 2017 |
| Abstract : Freeman_2017_J.Pharmacol.Exp.Ther_362_306 |
| ESTHER : Freeman_2017_J.Pharmacol.Exp.Ther_362_306 |
| PubMedSearch : Freeman_2017_J.Pharmacol.Exp.Ther_362_306 |
| PubMedID: 28576974 |
| Gene_locus related to this paper: human-LCAT |
| Title : Small molecule activation of lecithin cholesterol acyltransferase modulates lipoprotein metabolism in mice and hamsters - Chen_2012_Metabolism_61_470 |
| Author(s) : Chen Z , Wang SP , Krsmanovic ML , Castro-Perez J , Gagen K , Mendoza V , Rosa R , Shah V , He T , Stout SJ , Geoghagen NS , Lee SH , McLaren DG , Wang L , Roddy TP , Plump AS , Hubbard BK , Sinz CJ , Johns DG |
| Ref : Metabolism , 61 :470 , 2012 |
| Abstract : Chen_2012_Metabolism_61_470 |
| ESTHER : Chen_2012_Metabolism_61_470 |
| PubMedSearch : Chen_2012_Metabolism_61_470 |
| PubMedID: 22001333 |