N-acetylserotonin
N-Acetylserotonin is an intermediate in the metabolic pathway of melatonin and indoleamine in the pineal gland of mammalians. In most mammalian species the content of NAS (and melatonin) in the pineal gland shows clear circadian changes with the highest level occurring during the dark period. Experimental studies show that N-acetylserotonin possess free radical scavenging activity.
General
Type : Derivative of Tryptamine, Indole, Not AlphaBeta Hydrolase target, Natural
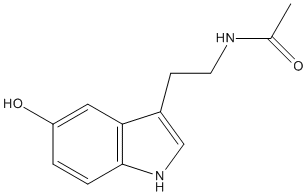
Chemical_Nomenclature : N-[2-(5-hydroxy-1H-indol-3-yl)ethyl]acetamide
Canonical SMILES : CC(=O)NCCC1=CNC2=C1C=C(C=C2)O
InChI : InChI=1S\/C12H14N2O2\/c1-8(15)13-5-4-9-7-14-12-3-2-10(16)6-11(9)12\/h2-3,6-7,14,16H,4-5H2,1H3,(H,13,15)
InChIKey : MVAWJSIDNICKHF-UHFFFAOYSA-N
Other name(s) : N-Acetyl-5-hydroxytryptamine || N-[2-(5-Hydroxy-1H-indol-3-yl)ethyl]acetamide || Normelatonin || CHEMBL33103 || CHEBI:17697 || ZINC66104 || DB04275 || N-acetylserotonine || Desmethylmelatonin
MW : 218.25
Formula : C12H14N2O2
CAS_number : 1210-83-9
PubChem : 903
UniChem : MVAWJSIDNICKHF-UHFFFAOYSA-N
Target
Structures : No structure
Families : Arylacetamide_deacetylase
References (1)
| Title : In vitro deacetylation of N-acetylserotonin by arylacetamide deacetylase - Huang_2023_J.Pineal.Res_75_e12870 |
| Author(s) : Huang Z , Li Y , Konishi K , Sakai Y , Tashiro K , Fukami T , Borjigin J |
| Ref : J Pineal Res , 75 :e12870 , 2023 |
| Abstract : |
| PubMedSearch : Huang_2023_J.Pineal.Res_75_e12870 |
| PubMedID: 37002641 |