Benzisothiazolinone-13
IC50 34.1 nM. Protects neuronal cells from oxidative stimuli and increased 2-AG levels in mouse brain
General
Type : Thiazolinone, Sulfur Compound
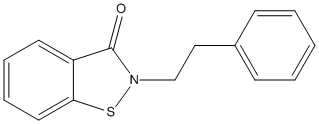
Chemical_Nomenclature : 2-(2-phenylethyl)-1,2-benzothiazol-3-one
Canonical SMILES : C1=CC=C(C=C1)CCN2C(=O)C3=CC=CC=C3S2
InChI : InChI=1S\/C15H13NOS\/c17-15-13-8-4-5-9-14(13)18-16(15)11-10-12-6-2-1-3-7-12\/h1-9H,10-11H2
InChIKey : VUMUTUGUKRGYGW-UHFFFAOYSA-N
Other name(s) : BTZ-13 || CHEMBL2391916 || SCHEMBL6550955 || BDBM50435396 || 2-Phenethyl-1,2-benzisothiazol-3(2H)-one
Target
Families : Benzisothiazolinone-13 ligand of proteins in family
Monoglyceridelipase_lysophospholip
Protein :
human-MGLL
References (1)
| Title : Benzisothiazolinone derivatives as potent allosteric monoacylglycerol lipase inhibitors that functionally mimic sulfenylation of regulatory cysteines - Castelli_2020_J.Med.Chem_63_1261 |
| Author(s) : Castelli R , Scalvini L , Vacondio F , Lodola A , Anselmi M , Vezzosi S , Carmi C , Bassi M , Ferlenghi F , Rivara S , Moller IR , Rand KD , Daglian J , Wei D , Dotsey EY , Ahmed F , Jung KM , Stella N , Singh S , Mor M , Piomelli D |
| Ref : Journal of Medicinal Chemistry , 63 :1261 , 2020 |
| Abstract : |
| PubMedSearch : Castelli_2020_J.Med.Chem_63_1261 |
| PubMedID: 31714779 |