BChE-IN-3
A87-7BO4 is the adduct of the inhibitor in the structure 7BO4 || BChE IC50 = 56.9 nM
General
Type : Derivative of Tryptophan,Indole,Carbamate,Cycloheptyl,Derivative of Tryptamine
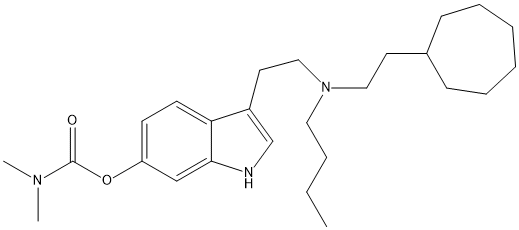
Chemical_Nomenclature : [3-[2-[butyl(2-cycloheptylethyl)amino]ethyl]-1H-indol-6-yl] N,N-dimethylcarbamate
Canonical SMILES : CCCCN(CCC1CCCCCC1)CCC2=CNC3=C2C=CC(=C3)OC(=O)N(C)C
InChI : InChI=1S\/C26H41N3O2\/c1-4-5-16-29(17-14-21-10-8-6-7-9-11-21)18-15-22-20-27-25-19-23(12-13-24(22)25)31-26(30)28(2)3\/h12-13,19-21,27H,4-11,14-18H2,1-3H3
InChIKey : DNYFOFJVAKFTIO-UHFFFAOYSA-N
Other name(s) : compound 45a,CHEMBL5171829,BDBM50604403,HY-144689,CS-0433866
MW : 427.6
Formula : C26H41N3O2
CAS_number :
PubChem : 163409034
UniChem : DNYFOFJVAKFTIO-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
Families : BChE-IN-3 ligand of proteins in family: BCHE
Stucture : 7BO4 Human Butyrylcholinesterase in complex with 3-(2-(butyl(2-cycloheptylethyl)amino)ethyl)-1H-indol-6-ol
Protein : human-BCHE
References (1)
| Title : From tryptophan-based amides to tertiary amines: Optimization of a butyrylcholinesterase inhibitor series - Meden_2022_Eur.J.Med.Chem_230_114248 |
| Author(s) : Meden A , Knez D , Brazzolotto X , Nachon F , Dias J , Svete J , Stojan J , Groselj U , Gobec S |
| Ref : Eur Journal of Medicinal Chemistry , 230 :114248 , 2022 |
| Abstract : Meden_2022_Eur.J.Med.Chem_230_114248 |
| ESTHER : Meden_2022_Eur.J.Med.Chem_230_114248 |
| PubMedSearch : Meden_2022_Eur.J.Med.Chem_230_114248 |
| PubMedID: 35299116 |
| Gene_locus related to this paper: human-BCHE |