Morpholino-Talinolol
Derivative of Talinolol unselective antagonist of the beta adrenergic receptors (betaAR). Much better inhibitor of soluble epoxide hydrolase than talinolol IC50 0.077+/-0.004 muM
General
Type : Urea derivative, Not A\/B H target, Adrenergic-Receptor-ligand, Morpholine, tert-Butyl
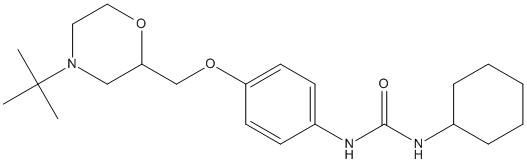
Chemical_Nomenclature : 1-[4-[[(2S)-4-tert-butylmorpholin-2-yl]methoxy]phenyl]-3-cyclohexylurea
Canonical SMILES : CC(C)(C)N1CCOC(C1)COC2=CC=C(C=C2)NC(=O)NC3CCCCC3
InChI : InChI=1S\/C22H35N3O3\/c1-22(2,3)25-13-14-27-20(15-25)16-28-19-11-9-18(10-12-19)24-21(26)23-17-7-5-4-6-8-17\/h9-12,17,20H,4-8,13-16H2,1-3H3,(H2,23,24,26)\/t20-\/m0\/s1
InChIKey : PSVUAGRHPQWOOO-FQEVSTJZSA-N
Other name(s) : G3T
MW : 389.53
Formula : C22H35N3O3
CAS_number :
PubChem : 138454247
UniChem : PSVUAGRHPQWOOO-FQEVSTJZSA-N
Target
Families : Morpholino-Talinolol ligand of proteins in family
Epoxide_hydrolase
Structure :
6HGX
Protein :
human-EPHX2
References (1)
| Title : Computer-Aided Selective Optimization of Side Activities of Talinolol - Hiesinger_2019_ACS.Med.Chem.Lett_10_899 |
| Author(s) : Hiesinger K , Kramer JS , Achenbach J , Moser D , Weber J , Wittmann SK , Morisseau C , Angioni C , Geisslinger G , Kahnt AS , Kaiser A , Proschak A , Steinhilber D , Pogoryelov D , Wagner K , Hammock BD , Proschak E |
| Ref : ACS Med Chem Lett , 10 :899 , 2019 |
| Abstract : |
| PubMedSearch : Hiesinger_2019_ACS.Med.Chem.Lett_10_899 |
| PubMedID: 31223445 |
| Gene_locus related to this paper: human-EPHX2 |