UW-MD-71
excellent selectivity profiles over both the three other HRs as well as butyrylcholinesterase (BChE) shows high and balanced in vitro affinities at both H3R and AChE with IC50 of 33.9nM and hH3R antagonism with Ki of 76.2nM, respectively
General
Type : Piperidine, Quinazoline, Multitarget, Histamine-Receptor3-H3-Antagonist
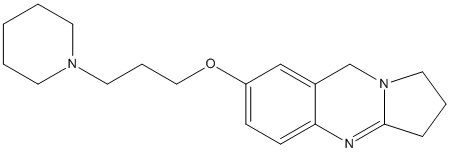
Chemical_Nomenclature : (7-(3-(piperidin-1-yl)propoxy)-1,2,3,9-tetrahydropyrrolo[2,1-b]quinazoline
Canonical SMILES : C1CCN(CC1)CCCOC2=CC3=C(C=C2)N=C4CCCN4C3
InChI : InChI=1S\/C19H27N3O\/c1-2-9-21(10-3-1)11-5-13-23-17-7-8-18-16(14-17)15-22-12-4-6-19(22)20-18\/h7-8,14H,1-6,9-13,15H2
InChIKey : UVYNECUSQBZMSD-UHFFFAOYSA-N
Other name(s) : CHEMBL3323035 || BDBM50053435
MW : 313.44
Formula : C19H27N3O
CAS_number :
PubChem : 118710487
UniChem : UVYNECUSQBZMSD-UHFFFAOYSA-N
Target
Families : UW-MD-71 ligand of proteins in family
ACHE
BCHE
Cholinesterase
References (3)
| Title : The dual-acting AChE inhibitor and H3 receptor antagonist UW-MD-72 reverses amnesia induced by scopolamine or dizocilpine in passive avoidance paradigm in rats - Sadek_2016_Physiol.Behav_165_383 |
| Author(s) : Sadek B , Khan N , Darras FH , Pockes S , Decker M |
| Ref : Physiol Behav , 165 :383 , 2016 |
| Abstract : |
| PubMedSearch : Sadek_2016_Physiol.Behav_165_383 |
| PubMedID: 27568232 |
| Title : The dual-acting H3 receptor antagonist and AChE inhibitor UW-MD-71 dose-dependently enhances memory retrieval and reverses dizocilpine-induced memory impairment in rats - Khan_2015_Behav.Brain.Res_297_155 |
| Author(s) : Khan N , Saad A , Nurulain SM , Darras FH , Decker M , Sadek B |
| Ref : Behavioural Brain Research , 297 :155 , 2015 |
| Abstract : |
| PubMedSearch : Khan_2015_Behav.Brain.Res_297_155 |
| PubMedID: 26467607 |
| Title : Synthesis, Biological Evaluation, and Computational Studies of Tri- and Tetracyclic Nitrogen-Bridgehead Compounds as Potent Dual-Acting AChE Inhibitors and hH3 Receptor Antagonists - Darras_2014_ACS.Chem.Neurosci_5_225 |
| Author(s) : Darras FH , Pockes S , Huang G , Wehle S , Strasser A , Wittmann HJ , Nimczick M , Sotriffer CA , Decker M |
| Ref : ACS Chem Neurosci , 5 :225 , 2014 |
| Abstract : |
| PubMedSearch : Darras_2014_ACS.Chem.Neurosci_5_225 |
| PubMedID: 24422467 |