Triazole-annulated-oxime-3A
General
Type : Bispyridinium, Bisoxime, Pyridinium, Triazol, Oxime
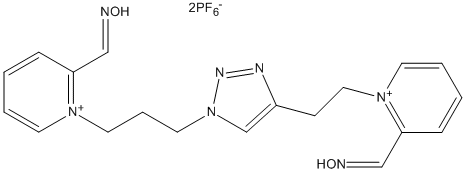
Chemical_Nomenclature : 2-((Hydroxyimino)methyl)-1-(3-(4-(2-(2-((hydroxyimino)methyl)pyridinium-1-yl)ethyl)-1H-1,2,3-triazol-1-yl)propyl)pyridinium
Canonical SMILES : C1=CC(=C[N+]([H])=O)N(C=C1)CCC[N]2C=C(N=N2)CCN3C=CC=CC3=C[N+]([H])=O
InChI : InChI=1S\/C19H21N7O2\/c27-20-14-18-6-1-3-9-24(18)11-5-12-26-16-17(22-23-26)8-13-25-10-4-2-7-19(25)15-21-28\/h1-4,6-7,9-10,14-16H,5,8,11-13H2\/p+2
InChIKey : YLYRUDRAEYQORF-UHFFFAOYSA-P
Other name(s) :
Target
Structure : No structure
Families : No family
References (2)
| Title : Click-chemistry-derived oxime library reveals efficient reactivators of nerve agent-inhibited butyrylcholinesterase suitable for pseudo-catalytic bioscavenging - Cadez_2025_Arch.Toxicol__ |
| Author(s) : Cadez T , Macek Hrvat N , Sinko G , Kalisiak J , Radic Z , Fokin VV , Sharpless KB , Taylor P , Kovarik Z |
| Ref : Archives of Toxicology , : , 2025 |
| Abstract : |
| PubMedSearch : Cadez_2025_Arch.Toxicol__ |
| PubMedID: 40032685 |
| Title : Reversal of Tabun Toxicity Enabled by a Triazole-Annulated Oxime Library-Reactivators of Acetylcholinesterase - Kovarik_2019_Chemistry_25_4100 |
| Author(s) : Kovarik Z , Kalisiak J , Macek Hrvat N , Katalinic M , Zorbaz T , Zunec S , Green C , Radic Z , Fokin VV , Sharpless KB , Taylor P |
| Ref : Chemistry , 25 :4100 , 2019 |
| Abstract : |
| PubMedSearch : Kovarik_2019_Chemistry_25_4100 |
| PubMedID: 30458057 |