Remdesivir
General
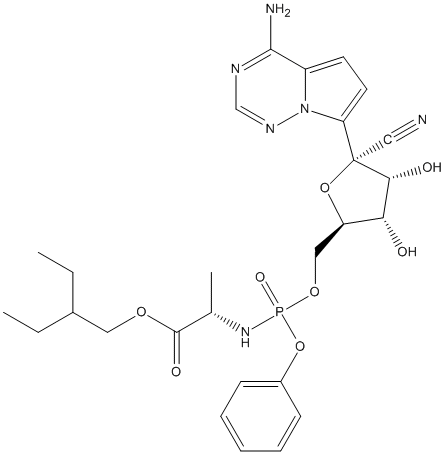
Type : Pro-Drug, Drug, Not A\/B H target, Propionate, Triazine
Chemical_Nomenclature : 2-ethylbutyl (2S)-2-[[[(2R,3S,4R,5R)-5-(4-aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-5-cyano-3,4-dihydroxyoxolan-2-yl]methoxy-phenoxyphosphoryl]amino]propanoate
Canonical SMILES : CCC(CC)COC(=O)C(C)NP(=O)(OCC1C(C(C(O1)(C#N)C2=CC=C3N2N=CN=C3N)O)O)OC4=CC=CC=C4
InChI : InChI=1S\/C27H35N6O8P\/c1-4-18(5-2)13-38-26(36)17(3)32-42(37,41-19-9-7-6-8-10-19)39-14-21-23(34)24(35)27(15-28,40-21)22-12-11-20-25(29)30-16-31-33(20)22\/h6-12,16-18,21,23-24,34-35H,4-5,13-14H2,1-3H3,(H,32,37)(H2,29,30,31)\/t17-,21+,23+,24+,27-,42-\/m0\/s1
InChIKey : RWWYLEGWBNMMLJ-YSOARWBDSA-N
Other name(s) : 3QKI37EEHE || GS 5734 || GS-5734 || CHEMBL4065616 || SCHEMBL17712225 || CHEBI:145994
MW : 602.6
Formula : C27H35N6O8P
CAS_number : 1809249-37-3
PubChem : 121304016
UniChem : RWWYLEGWBNMMLJ-YSOARWBDSA-N
Target
Structures : No structure
Families : Carb_B_Chordata, Carboxypeptidase_S10
References (8)
| Title : Assessing in vitro stability of remdesivir (GS-5734) and conversion to GS-441524 in feline plasma and whole blood - Coggins_2024_Vet.Q_44_1 |
| Author(s) : Coggins SJ , Kimble B , Malik R , Thompson MF , Norris JM , Govendir M |
| Ref : Vet Q , 44 :1 , 2024 |
| Abstract : |
| PubMedSearch : Coggins_2024_Vet.Q_44_1 |
| PubMedID: 38288972 |
| Title : Key Metabolic Enzymes Involved in Remdesivir Activation in Human Lung Cells - Li_2021_Antimicrob.Agents.Chemother__AAC0060221 |
| Author(s) : Li R , Liclican A , Xu Y , Pitts J , Niu C , Zhang J , Kim C , Zhao X , Soohoo D , Babusis D , Yue Q , Ma B , Murray BP , Subramanian R , Xie X , Zou J , Bilello JP , Li L , Schultz BE , Sakowicz R , Smith BJ , Shi PY , Murakami E , Feng JY |
| Ref : Antimicrobial Agents & Chemotherapy , :AAC0060221 , 2021 |
| Abstract : |
| PubMedSearch : Li_2021_Antimicrob.Agents.Chemother__AAC0060221 |
| PubMedID: 34125594 |
| Title : Comparison of anti-SARS-CoV-2 activity and intracellular metabolism of remdesivir and its parent nucleoside - Tao_2021_Curr.Res.Pharmacol.Drug.Discov_2_100045 |
| Author(s) : Tao S , Zandi K , Bassit L , Ong YT , Verma K , Liu P , Downs-Bowen JA , McBrayer T , LeCher JC , Kohler JJ , Tedbury PR , Kim B , Amblard F , Sarafianos SG , Schinazi RF |
| Ref : Curr Res Pharmacol Drug Discov , 2 :100045 , 2021 |
| Abstract : |
| PubMedSearch : Tao_2021_Curr.Res.Pharmacol.Drug.Discov_2_100045 |
| PubMedID: 34870151 |
| Title : Remdesivir potently inhibits carboxylesterase-2 through covalent modifications: signifying strong drug-drug interactions - Shen_2021_Fundam.Clin.Pharmacol_35_432 |
| Author(s) : Shen Y , Eades W , Yan B |
| Ref : Fundamental & Clinical Toxicology , 35 :432 , 2021 |
| Abstract : |
| PubMedSearch : Shen_2021_Fundam.Clin.Pharmacol_35_432 |
| PubMedID: 33369768 |
| Gene_locus related to this paper: human-CES2 |
| Title : Comparative therapeutic efficacy of remdesivir and combination lopinavir, ritonavir, and interferon beta against MERS-CoV - Sheahan_2020_Nat.Commun_11_222 |
| Author(s) : Sheahan TP , Sims AC , Leist SR , Schafer A , Won J , Brown AJ , Montgomery SA , Hogg A , Babusis D , Clarke MO , Spahn JE , Bauer L , Sellers S , Porter D , Feng JY , Cihlar T , Jordan R , Denison MR , Baric RS |
| Ref : Nat Commun , 11 :222 , 2020 |
| Abstract : |
| PubMedSearch : Sheahan_2020_Nat.Commun_11_222 |
| PubMedID: 31924756 |
| Title : Remdesivir: A Review of Its Discovery and Development Leading to Emergency Use Authorization for Treatment of COVID-19 - Eastman_2020_ACS.Cent.Sci_6_672 |
| Author(s) : Eastman RT , Roth JS , Brimacombe KR , Simeonov A , Shen M , Patnaik S , Hall MD |
| Ref : ACS Cent Sci , 6 :672 , 2020 |
| Abstract : |
| PubMedSearch : Eastman_2020_ACS.Cent.Sci_6_672 |
| PubMedID: 32483554 |
| Title : Coronavirus Susceptibility to the Antiviral Remdesivir (GS-5734) Is Mediated by the Viral Polymerase and the Proofreading Exoribonuclease - Agostini_2018_mBio_9_ |
| Author(s) : Agostini ML , Andres EL , Sims AC , Graham RL , Sheahan TP , Lu X , Smith EC , Case JB , Feng JY , Jordan R , Ray AS , Cihlar T , Siegel D , Mackman RL , Clarke MO , Baric RS , Denison MR |
| Ref : MBio , 9 : , 2018 |
| Abstract : |
| PubMedSearch : Agostini_2018_mBio_9_ |
| PubMedID: 29511076 |
| Title : Therapeutic efficacy of the small molecule GS-5734 against Ebola virus in rhesus monkeys - Warren_2016_Nature_531_381 |
| Author(s) : Warren TK , Jordan R , Lo MK , Ray AS , Mackman RL , Soloveva V , Siegel D , Perron M , Bannister R , Hui HC , Larson N , Strickley R , Wells J , Stuthman KS , Van Tongeren SA , Garza NL , Donnelly G , Shurtleff AC , Retterer CJ , Gharaibeh D , Zamani R , Kenny T , Eaton BP , Grimes E , Welch LS , Gomba L , Wilhelmsen CL , Nichols DK , Nuss JE , Nagle ER , Kugelman JR , Palacios G , Doerffler E , Neville S , Carra E , Clarke MO , Zhang L , Lew W , Ross B , Wang Q , Chun K , Wolfe L , Babusis D , Park Y , Stray KM , Trancheva I , Feng JY , Barauskas O , Xu Y , Wong P , Braun MR , Flint M , McMullan LK , Chen SS , Fearns R , Swaminathan S , Mayers DL , Spiropoulou CF , Lee WA , Nichol ST , Cihlar T , Bavari S |
| Ref : Nature , 531 :381 , 2016 |
| Abstract : |
| PubMedSearch : Warren_2016_Nature_531_381 |
| PubMedID: 26934220 |