JZP-430
General
Type : Carbamate, Morpholine, Sulfur Compound, Thiadiazol
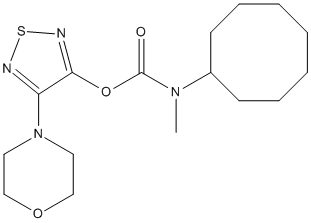
Chemical_Nomenclature : (4-morpholin-4-yl-1,2,5-thiadiazol-3-yl) N-cyclooctyl-N-methylcarbamate
Canonical SMILES : CN(C1CCCCCCC1)C(=O)OC2=NSN=C2N3CCOCC3
InChI : InChI=1S\/C16H26N4O3S\/c1-19(13-7-5-3-2-4-6-8-13)16(21)23-15-14(17-24-18-15)20-9-11-22-12-10-20\/h13H,2-12H2,1H3
InChIKey : WKSHMJCYWFOADB-UHFFFAOYSA-N
Other name(s) : (4-Morpholin-4-yl-1,2,5-thiadiazol-3-yl) N-cyclooctyl-N-methylcarbamate || 4-(4-Morpholinyl)-1,2,5-thiadiazol-3-ylN-cyclooctyl-N-methylcarbamate || CHEMBL3613671 || SCHEMBL22799434 || JZP 430 || ZINC473127446
MW : 354.5
Formula : C16H26N4O3S
CAS_number : 1672691-74-5
PubChem : 121513897
UniChem : WKSHMJCYWFOADB-UHFFFAOYSA-N
Target
Families : JZP-430 ligand of proteins in family
ABHD6-Lip
Protein :
human-ABHD6
References (3)
| Title : Optimization of 1,2,5-thiadiazole carbamates as potent and selective ABHD6 inhibitors - Patel_2015_ChemMedChem_10_253 |
| Author(s) : Patel JZ , Nevalainen TJ , Savinainen JR , Adams Y , Laitinen T , Runyon RS , Vaara M , Ahenkorah S , Kaczor AA , Navia-Paldanius D , Gynther M , Aaltonen N , Joharapurkar AA , Jain MR , Haka AS , Maxfield FR , Laitinen JT , Parkkari T |
| Ref : ChemMedChem , 10 :253 , 2015 |
| Abstract : |
| PubMedSearch : Patel_2015_ChemMedChem_10_253 |
| PubMedID: 25504894 |
| Gene_locus related to this paper: human-ABHD6 |
| Title : Optimization of 1,2,5-thiadiazole carbamates as potent and selective ABHD6 inhibitors - Patel_2015_ChemMedChem_10_253 |
| Author(s) : Patel JZ , Nevalainen TJ , Savinainen JR , Adams Y , Laitinen T , Runyon RS , Vaara M , Ahenkorah S , Kaczor AA , Navia-Paldanius D , Gynther M , Aaltonen N , Joharapurkar AA , Jain MR , Haka AS , Maxfield FR , Laitinen JT , Parkkari T |
| Ref : ChemMedChem , 10 :253 , 2015 |
| Abstract : |
| PubMedSearch : Patel_2015_ChemMedChem_10_253 |
| PubMedID: 25504894 |
| Gene_locus related to this paper: human-ABHD6 |
| Title : Optimization of 1,2,5-thiadiazole carbamates as potent and selective ABHD6 inhibitors - Patel_2015_ChemMedChem_10_253 |
| Author(s) : Patel JZ , Nevalainen TJ , Savinainen JR , Adams Y , Laitinen T , Runyon RS , Vaara M , Ahenkorah S , Kaczor AA , Navia-Paldanius D , Gynther M , Aaltonen N , Joharapurkar AA , Jain MR , Haka AS , Maxfield FR , Laitinen JT , Parkkari T |
| Ref : ChemMedChem , 10 :253 , 2015 |
| Abstract : |
| PubMedSearch : Patel_2015_ChemMedChem_10_253 |
| PubMedID: 25504894 |
| Gene_locus related to this paper: human-ABHD6 |