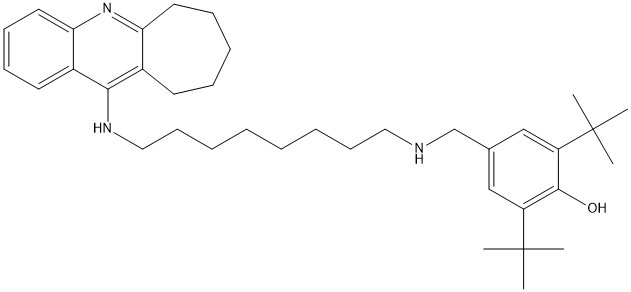
Di-tert-butyl-phenol-octylamino-cycloheptaquinolin-18d
BChE IC50 5.96 +/- 0.58 nM
General
Type : Multitarget,Derivative of Tacrine,Quinoline,Antioxidant,tert-Butyl,Alkyl linked bis-ligand
Chemical_Nomenclature : 2,6-Di-tert-butyl-4-{[8-(7,8,9,10-tetrahydro-6H-cyclohepta[b]quinolin-11-ylamino)-octylamino]-methyl}-phenol
Canonical SMILES : C1=CC=C2C(=C1)N=C4C(=C2NCCCCCCCCNCC3=CC(=C(C(=C3)C(C)(C)C)O)C(C)(C)C)CCCCC4
InChI : InChI=1S\/C37H55N3O\/c1-36(2,3)30-24-27(25-31(35(30)41)37(4,5)6)26-38-22-16-9-7-8-10-17-23-39-34-28-18-12-11-13-20-32(28)40-33-21-15-14-19-29(33)34\/h14-15,19,21,24-25,38,41H,7-13,16-18,20,22-23,26H2,1-6H3,(H,39,40)
InChIKey : IWMFLEQRODEMEX-UHFFFAOYSA-N
Other name(s) : 18d
MW : 557.86
Formula : C37H55N3O
CAS_number :
PubChem :
UniChem : IWMFLEQRODEMEX-UHFFFAOYSA-N
IUPHAR :
Wikipedia :
Target
Families : Di-tert-butyl-phenol-octylamino-cycloheptaquinolin-18d ligand of proteins in family: BCHE
Stucture :
Protein : human-BCHE
References (1)
| Title : Combining Experimental and Computational Methods to Produce Conjugates of Anticholinesterase and Antioxidant Pharmacophores with Linker Chemistries Affecting Biological Activities Related to Treatment of Alzheimer's Disease - Makhaeva_2024_Molecules_29_321 |
| Author(s) : Makhaeva GF , Kovaleva NV , Rudakova EV , Boltneva NP , Lushchekina SV , Astakhova TY , Timokhina EN , Serkov IV , Proshin AN , Soldatova YV , Poletaeva DA , Faingold, II , Mumyatova VA , Terentiev AA , Radchenko EV , Palyulin VA , Bachurin SO , Richardson RJ |
| Ref : Molecules , 29 :321 , 2024 |
| Abstract : Makhaeva_2024_Molecules_29_321 |
| ESTHER : Makhaeva_2024_Molecules_29_321 |
| PubMedSearch : Makhaeva_2024_Molecules_29_321 |
| PubMedID: 38257233 |